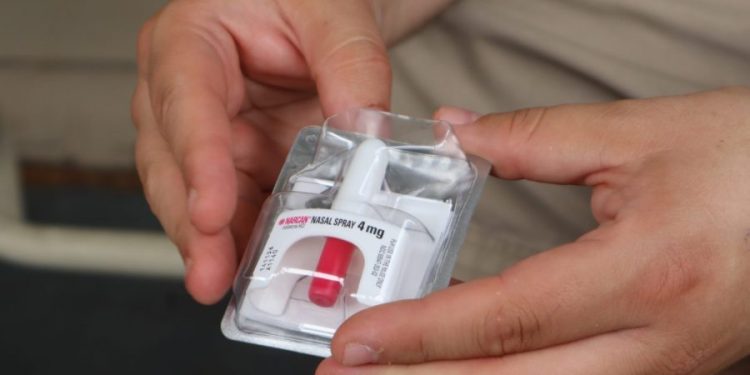
The anti-opioid overdose drug Narcan should be made available for over-the-counter use, advisers to the Food and Drug Administration said Wednesday.
A joint FDA advisory panel unanimously voted 19-0 to recommend the agency approve the drug, which is currently only available by prescription. FDA approval would allow more people to acquire the treatment more easily in more places.
Naloxone is widely viewed as the best and possibly only treatment for opioid overdoses, though its status as a prescription-only medication has been a major barrier.
Advocates have said broadening community access to the medication could also help remove the stigma associated with it.
Every state has “standing orders” that are intended to make the drug available at the pharmacy without a prescription, but not all pharmacies carry it and the process can be complicated for pharmacists.
The FDA will make a decision by March 29, though panel members said it should be done sooner in order to substantially increase access to the lifesaving drug.
The recommendation is not binding, but the agency is expected to accept it. FDA pushed drug manufacturers to submit applications for a nonprescription version of naloxone last fall, and granted fast-track status to the two companies that applied.
Narcan, the brand-name nasal spray manufactured by Emergent BioSolutions, is the most commonly sold emergency treatment for opioid overdose in United States pharmacies. It was first approved in 2015 to treat known or suspected opioid overdoses for people of all ages, including newborns.
Advisory panel members broadly agreed there was little to no risk involved in making Narcan available over-the-counter, even if it’s given to someone who is not experiencing an overdose.
However, there were some concerns whether Narcan’s labeling and instructions were clear enough to avoid user errors.
For instance, the company cited examples during its trials of people failing to wait the recommended two to three minutes between doses if the individual remains unresponsive. There were also reported instances of people spraying Narcan into the air, rather than into an overdose victim.
But the panelists said any of the label issues shouldn’t slow FDA approval, and the opioid epidemic warrants an urgent response.
“There’s no reason to keep this as a prescription, let’s get it out there and save some lives,” said Elizabeth Coykendall, a paramedic in North Carolina and temporary voting member of the panel.
Matt Harwig, a spokesman for Emergent BioSolutions, said the cost of the drug hadn’t been decided. Harm reduction advocates have expressed concern that it could be priced out of reach for groups that distribute it to people who need it the most.