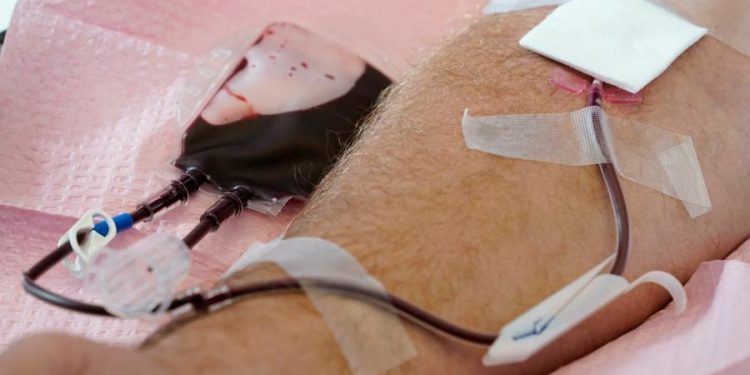
The Food and Drug Administration (FDA) on Thursday loosened restrictions on blood donations by men who have sex with men, a change that could ease blood shortages by allowing more people to donate.
The agency said it will recommend a series of “individual risk-based questions” that will be the same for every donor, regardless of sexual orientation, sex or gender. Gay and bisexual men in monogamous relationships will be allowed to donate blood.
“The implementation of these recommendations will represent a significant milestone for the agency and the LGBTQI+ community,” said Peter Marks, director of the FDA’s Center for Biologics Evaluation and Research.
Under current FDA guidelines for donating blood, men who have sex with men are permitted to donate blood after a three-month deferral period in which they abstain from sexual encounters with men. This change was made in 2020, after the previous guidance had mandated a 12-month deferral period.
The new policy eliminates current time-based restrictions in favor of a more inclusive policy the agency said was based on the best scientific evidence, while also maintaining the safety of the blood supply.
“The FDA has worked diligently to evaluate our policies and ensure we had the scientific evidence to support individual risk assessment for donor eligibility while maintaining appropriate safeguards to protect recipients of blood products,” Marks said.
Under the final guidance issued Thursday, all prospective blood donors will answer a series of individual, risk-based questions to determine eligibility.
All prospective donors who report having a new sexual partner, or more than one sexual partner in the past three months, and anal sex in the past three months, would be deferred for three months to reduce the likelihood of donations by individuals with new or recent HIV infection, FDA said.